Morphological basis of dental implantation
Machine translation
Original article is written in RU language (link to read it) .
Dental implantation is a modern technology that allows you to implant an artificial root into the bone tissue of the patient’s jaw. The implant design consists of a titanium screw and abutment. Subsequently, implants are used as a support for orthopedic structures. The abutment is installed inside the titanium screw only after the latter has healed.
Placement of the implant is accompanied by trauma to the patient’s bone tissue, then regeneration processes are launched, which lead to the formation of first reticulofibrous and subsequently lamellar bone tissue.
Learn more about the factors influencing implant healing in the webinar Single-stage implantation with immediate loading .
Clinically, normal bone regeneration should lead to direct tight contact between bone tissue and the implant, in other words, implant healing is equal to osseointegration.
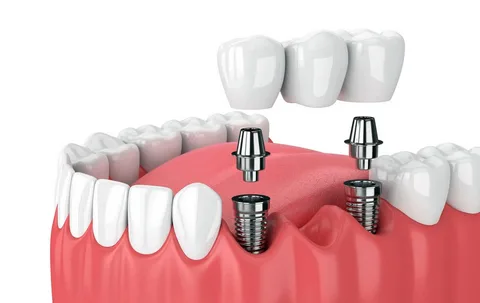
Figure 1. Dental implantation.
It was previously believed that the fibrous tissue surrounding it plays a major role in the osseointegration of the implant. Scientists assumed that collagen fibers begin to gradually grow into it, due to which the implant is connected through fibrous tissue to the bone, similar to Sharpey’s fibers.
The use of flat designs of Linkow and Weiss implants is based on fibrous implantation. In this regard, their surface was equipped with irregularities, bends and holes that allowed bone tissue to grow through the implant.
If the healing process is unfavorable or if excessive premature load is placed on the implant, a thick layer of connective tissue forms between it and the alveolar bone and disintegration occurs. Fibrous tissue cannot be considered as a reliable support for an implant; the functioning of the latter with this support does not exceed ten years.
The engraftment of flat-shaped Linkow and Weiss implants is based on another mechanism - osteofibrous integration, which is based on partial direct contact formed due to the growth of bone tissue into the holes of the implant. In this case, the proportion of direct contact with bone tissue should be from 22% of the area of the implant body.
The type of integration is determined by the initial loads on the implant. With osteofibrous integration, the supporting layer has shock absorber characteristics due to the elastic fibrous capsule. Scientists were unable to experimentally prove the formation of fibers between the fibrous tissue of the implant and the bone surface. The successful healing of flat implant designs has been poorly studied. Moreover, some structures have been functioning for 25 years.
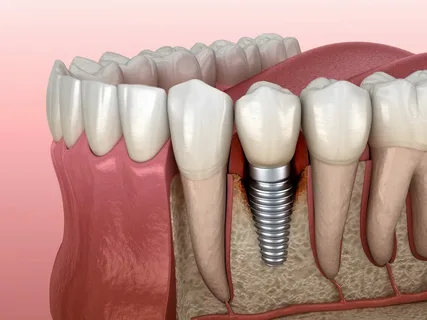
Figure 2. Osseointegration of the implant.
Stages of bone healing after implantation
A bone wound after dental implantation undergoes the same stages of healing that are characteristic of wound healing due to a jaw fracture. Healing patterns depend on the implant material and type of surgical procedure.
Healing of a bone wound includes the following stages:
inflammation,
proliferation,
healing.
Stage of inflammation
The beginning of the inflammation stage is associated with a reaction to foreign interference, the detection of a foreign body in the tissues, this is how the body perceives the implant. The duration of the inflammation stage is about 10 days, but its duration may increase.
Alteration
An implant placed in a bone bed causes alteration in the inflammation stage. Alteration is considered as a set of the following changes:
damage to cellular elements,
mobilization of cell defenses,
activation of the defenses of the entire body as a whole.
General characteristics of alteration processes
A set of morphological and biochemical changes.
Local vascular reactions.
Tissue necrosis is observed at the lesion site.
The body triggers reactions of integral regulatory systems, where an important role is played by vasoactive and chemotactic substances that determine the degree of inflammation.
Dystrophic disorders of cells and intercellular substance.
Proteins are absorbed onto the surface of the implants.
On the surface of the implant, platelet aggregation increases, coagulation of a cascade nature increases, cytokines are released, specific and nonspecific cellular reactions are activated.
Figure 3. Total oral rehabilitation through implantation.
It is difficult to identify early signs of exudation and alteration, since reparative processes predominate during the first week. The reactions that occur in tissues lead to the onset of tissue acidosis and an increase in osmotic pressure. At the border of bone tissue and implant, products accumulate that cause disruption of homeostasis and vascular permeability. The main criterion for the transition from alteration to the exudation stage is a violation of vascular permeability.
Exudation
General characteristics of exudation processes
Hemodynamics are impaired.
Arterial vascular hyperemia develops under the influence of inflammatory mediators, the outflow of blood and lymph is disrupted. An important role in the implementation of these processes belongs to histamine and serotonin.
Local thrombosis is observed.
Acidosis is noted, leading to an increase in the hydrophilicity of tissue colloids, and, as a consequence, collagen fibers, which contributes to a slowdown in blood flow in small vessels due to weakening of the walls.
A significant slowdown in blood flow can cause stasis.
The complement system is activated.
Exudation and its consequence, increased edema, perform a protective function, but lead to an increase in blood pressure in small vessels and osmotic pressure in surrounding tissues. The result is the movement of leukocytes into the area of damage. The migration of leukocytes is always directed to the area of tissue damage. If the course is favorable, on the third day after the implantation operation, blood circulation in the damaged tissues begins to be restored.
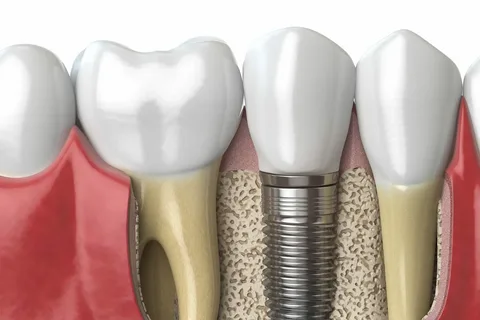
Figure 4. Intimate contact between bone and imalantate surface.
Proliferation stage
At this stage, cell differentiation is observed, tissue trophism is gradually restored, which is evidence of healing of the bone wound. The duration of this stage is about 6 weeks, but initial signs may appear as early as the third day after surgery.
The following processes are observed in the connective tissue adjacent to the implant:
neoplasm of blood vessels,
cell proliferation and differentiation,
activation of collagen synthesis,
movement of cellular elements to the site of inflammation.
Epithelialization of the wound, cell differentiation, growth and keratinization of the epithelium occur. The development of vascular anastomoses causes the restoration of microcirculation and an increase in oxygen saturation of tissues, which accelerates the flow of nutrients.
At the proliferation stage, the following reparative processes occur:
immature elements of connective tissue are transformed into young granulation tissue,
young scar tissue is formed,
Scar tissue is gradually rebuilt.
Regeneration stage
Regeneration processes are observed from the first days after implantation; they reach their maximum by the end of the month. Provided that no loads are applied to the implant body, bone tissue begins to form around it and its blood supply is established. The formed callus is represented by fibrocartilaginous elements, which is similar to endochondral ossification.
Establishing trophism and the flow of oxygen to the site of damage stimulates local hemodynamics and microcirculation, which has a beneficial effect on the reconstruction of bone tissue.
Bone mineralization is caused by the load on the implant. Without load, the bone tissue that is in direct contact with the implant has many medullary spaces, voids in the spongy substance. While densely formed bone tissue is determined around the loaded implant.
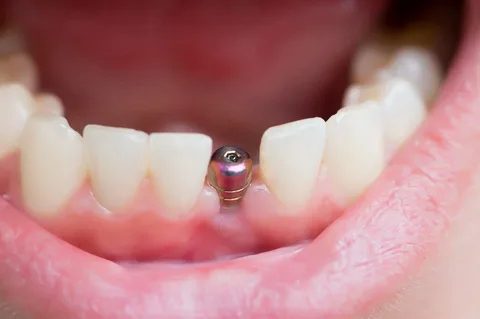
Figure 5. View in the oral cavity.
Cytogenesis of immature mesenchymal cells and their subsequent differentiation leads to the formation of a matrix, which consists of collagen, glycosaminoglycans, glycoproteins and glycolipids; this matrix undergoes restructuring. The immature future bone matrix remodels in response to implant loading as well as physiological bone recession. These mechanisms underlie the implant healing process.
If healing proceeds favorably, the implant is well adapted, adventitial cells differentiate on its surface and fill the areas of damaged bone. At the same time, processes of resorption and ossification are observed in bone tissue. The rate of mineralization of newly formed bone can reach 1 mm per day.
All modern technologies, methods and tools for achieving a stable result of implant treatment are collected in the online course Guided regeneration and navigational implantation .
