The clinical use of the Er,Cr:YSGG laser in endodontic therapy
Total elimination of bacteria from infected root-canal systems remains the most important objective of endodontic therapy. However, in spite of a plethora of new products and techniques, achieving this objective continues to elude our profession. Historically, endodontic treatment focused on root-canal disinfection with “entombment” of remaining bacteria within dentinal tubules and inaccessible areas of the root-canal system. Although many factors have been implicated in the aetiology of endodontic failures, it has become evident that these “entombed” bacteria play a pivotal role in the persistence of endodontic disease.
Although impressive results have been obtained in vitro, laser energy alone has not been able to achieve total bacterial kill in extracted teeth. From a clinical perspective, it is apparent that a combination of different treatment modalities is required to sterilise root-canal systems. In addition, many clinical obstacles exist that further complicate the clinician’s ability to achieve this goal. These include, but are not limited to: restricted endodontic access, complex root-canal anatomy, limitations of irrigation and instrumentation techniques, inability to entomb bacteria, and the inability to reach and eliminate bacteria deep within the tooth structure.
While the purpose of this article is to focus on the clinical use of the Er,Cr:YSGG laser with radial-firing tips, a definitive treatment protocol needs to be in place to reduce the intra-canal bacterial load prior to laser usage and to facilitate delivery of the laser energy to the most critical part of the root canal, the apical third.
The erbium, chromium-doped yttrium, scandium, gallium and garnet (Er,Cr:YSGG) laser emits at a wave-length of 2,780 nm and is highly absorbed by water. The lower the penetration depth in water or tissue (or the higher the absorption), the greater the ability of the laser to cut or ablate tissue (Fig. 1). Since this wavelength is very similar to the absorption maximum of water in hydroxyapatite, photo-ablation occurs where water evaporates instantaneously, thereby ablating the surrounding tissue. Gordon et al. found that it was possible to achieve expansion and collapse of intratubular water as deep as 1,000µm or more. This micro-pulse-induced absorption was capable of producing acoustic waves sufficiently strong to disrupt and kill intratubular bacteria.
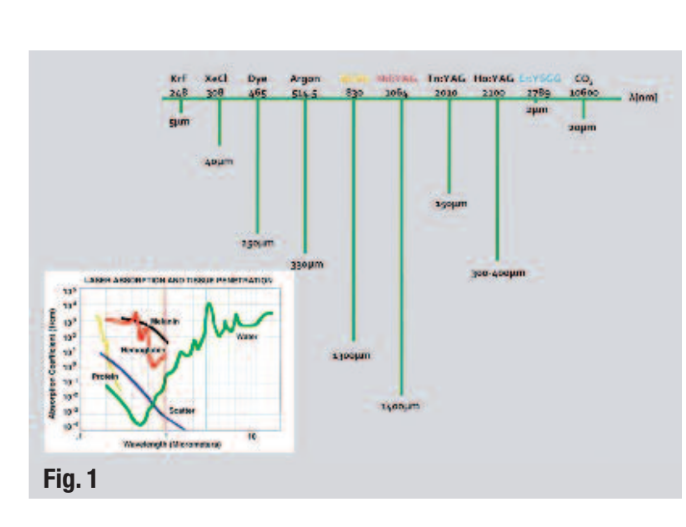
These findings are significant, as bacteria have been identified at depths of 1,000µm, with E. faecalis at depths of 800µm. Irrigants such as sodium hypochlorite have a limited effect on these bacteria with penetration depths of only 100µm. Increasing concentration, exposure time and temperature was recently found to improve NaOCl penetration.
Promising bacterial kill rates using the Er,Cr:YSGG laser with radial-firing tips have been reported in extracted teeth. A disinfection reduction of 99.7% was obtained for E. faecalis at depths of 200µm into dentine and 94.1 % (1 log) at depths of 1,000µm.
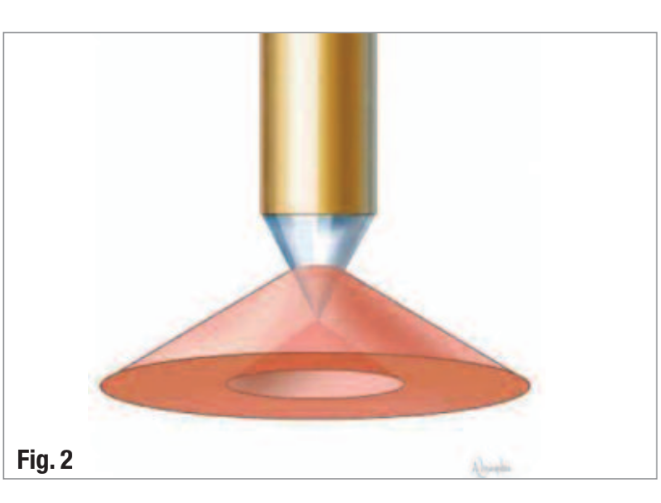
The development of the radial-firing laser tip (Biolase Technology, Inc.) with a tip shape that emits the laser energy as a broad cone allows better coverage of the root-canal walls than end-firing tips (Fig. 2). This facilitates entry of the emitted laser energy into the dentinal tubules reaching bacteria that have penetrated deep into the dentine.
Treatment protocol
Current techniques incorporating hand and/or rotary instrumentation, positive pressure irrigation, with or without sonic and ultrasonic agitation, fall short of total canal disinfection. Thetreatment protocol presented in this article incorporates three main components: management of the working width of the root canal, negative pressure apical irrigation and intra-canal laser therapy.
Working width management
The working width (WW) of a root canal is the diameter of the canal immediately before its apical constriction. Allen found that 97 % of canals not cleaned to their WW had residual debris in the critical apical region, while 100 % of those cleaned to their WW were free of debris 1 mm from the apical constriction. Studies have demonstrated that we need to clean to larger sizes to remove bacteria and debris. Conventional tapered files cannot accomplish this without transporting the canal, creating strip perforations, weakening the tooth or separating instruments. The LightSpeed LSX (Discus Dental) file is a unique, extremely flexible, taperless, NiTi instrument capable of cleaning to the WW. The final apical size is the instrument size that completes WW preparation and is determined when the LSX file binds 4 mm (or more) from the working length (WL), and requires a firm push to reach WL. The customised apical preparations created are critical for predictably successful endodontics and provide significant advantages:
- effective removal of infected material, debris, inflamed and necrotic tissue from the apical region;
- allows placement of irrigating needle to WL for negative pressure apical irrigation;
- facilitates placement of intra-canal medication deeper within the canal;
- and facilitates placement of radial-firing laser tip within 1 mm of WL.
Negative pressure apical irrigation
There are two main reasons that irrigants fail to reach the critical last 3 mm of a root canal. Firstly, using positive pressure irrigation with a side-vented needle there is little flushing beyond the depth of the needle. Most of the irrigant follows the path of least resistance and backs out of the canal, with apical flushing penetrating only 1 to 2 mm apical to the end of the needle. To achieve effective apical flushing, the needle tip needs to be placed 1 mm from WL, which dramatically increases the risk of a sodium-hypochlorite accident.
Secondly, the presence of apical vapour lock from air trapped in the canal, as well as ammonia and carbon dioxide released from the dissolving action of sodium hypochlorite on pulp tissue, prevents penetration of irrigants into the apical third. This vapour lock cannot be removed with hand or rotary files, sonic or ultrasonic activation. In a recent study, vapour lock resulted in “gross retention of debris and smear layer remnants” in the apical 0.5 to 1.0 mm of closed root-canal systems.
The EndoVac (Discus Dental) is a true apical negative pressure irrigating system that provides continuous, high-volume irrigation of fresh fluids to the canal terminus with simultaneous evacuation. It is comprised of a master delivery tip (Fig. 3) that delivers fluid to the pulp chamber and a macro- and micro-cannula (Fig. 4) that draw the fluid from the chamber to the canal terminus by way of evacuation.
This system eliminates vapour lock and provides superior cleaning, disinfecting and smear layer removal, while virtually eliminating the threat of sodium hypochlorite accident. When compared to positive pressure irrigation with a ProRinse needle, EndoVac produced canals that were 366 and 671% cleaner 1 and 3 mm from WL, respectively.
When EndoVac was used in combination with LightSpeed LSX instrumentation, canals were 99 and 99.5 % free of debris 1 and 3 mm from WL, respectively.
Intra-canal laser therapy
The final stage of root-canal preparation and disinfectioniscompletedwiththe Waterlase MDlaser (Er,Cr:YSGG) using radial-firing tips (Biolase Technology).
The laser tips are available in two sizes: RFT2 and RFT3 with diameters of 275 and 415µm, respectively (Fig. 5). The RFT2 tip is inserted 1 mm short of WL, requiring canal preparation sizes of ISO 30 or more while the RFT3 tip is inserted to the junction of middle and apical thirds, requiring canal sizes of ISO 45 or more. These sizes fall well within typical working width preparation sizes prepared with LSXfiles. Intracanal laser therapy is performed in two phases, the Cleaning Phase for smear layer and debris removal, and the Disinfection Phase for tissue ablation and bacterial elimination.

Cleaning phase (1.25 W; 50 Hz; 24 % air; 30 % water)
This phase uses water and removes smear layer and debris without using chemical irrigants. It takes two to three minutes per canal and uses Hydro-Photonics to create a powerful micro-agitation effect throughout the canal system.
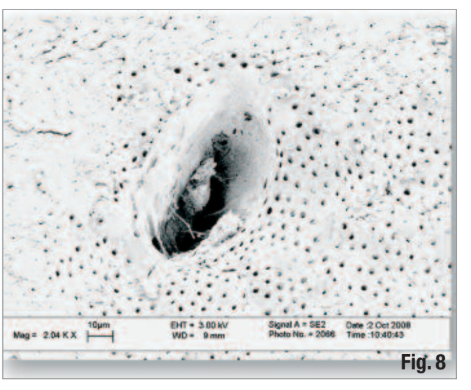
It is generally accepted that smear layer removal facilitates the cleaning and disinfecting of the dentinal tubules and improves the sealing of the root canal. When merging results of two studies, the Er,Cr:YSGG with radial-firing tips produced significantly better smear layer removal in the apical, middle and coronal thirds than two rotary techniques. This extremely efficient action opens the dentinal tubules, lateral canals and isthmuses in preparation for disinfection (Figs. 6–8).


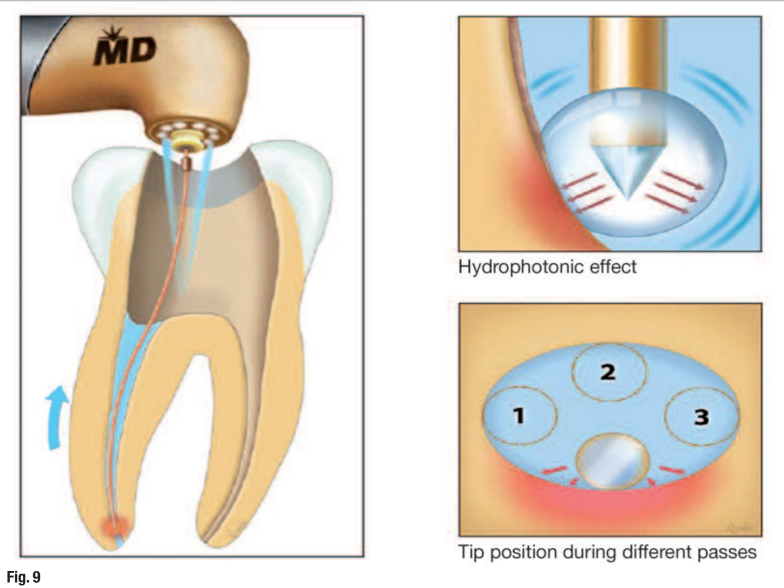
Technique for cleaning phase after completion of access, working width preparation and negative pressure irrigation:
- use the RFT2 to perform apical and partial coronal two-thirds cleaning;
- select the recommended laser settings in the wet mode;
- fill canal with sterile solution;
- insert RFT2 tip 1 mm short of WL;
- activate laser on withdrawal of tip coronally at approximately 1 mm/s and maintain tip in contact with the side surface of the canal wall during the entire apical to coronal pass;
- repeat steps 4 and 5 one or two more times to ensure that the entire inner canal has been cleaned (Fig. 9);
- place the RFT3 tip in handpiece to perform final cleaning of the coronal two-thirds;
- fill canal with sterile solution;
- insert the tip to the junction of apical and middle third of the root canal; and
- repeat steps 5 and 6.
Disinfection phase (0.75 W; 20 Hz; 10 %air; 0 %water)
As stated previously, the laser energy emitted from the Er,Cr:YSGG laser is highly absorbed by water in tissue and micro-organisms, resulting in instantaneous photo-ablation. In addition, the resulting micro-pulse expansion and collapse of intratubular water produce acoustic waves sufficiently strong to disrupt and kill intratubular bacteria. This effect is most effective in a dry mode, as the laser energy is not absorbed by the water spray and can exert its full effect on the bacteria. This was confirmed by Gordon et al., who achieved a 99.7 % kill rate for E. faecalis in the dry mode. Technique for the disinfection phase is the same as the cleaning phase but with different laser settings in the dry mode.
Clinical applications
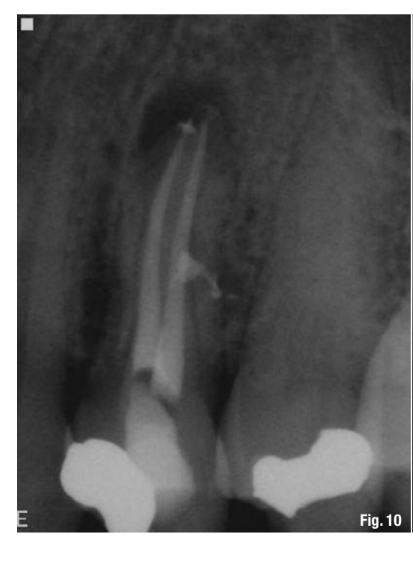
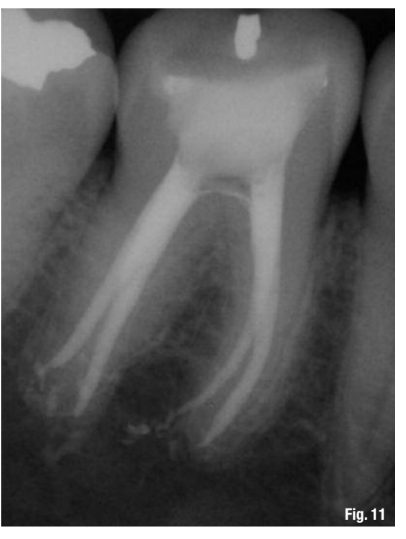
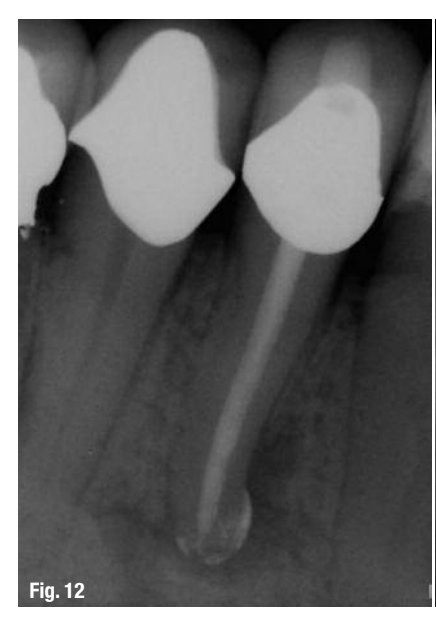
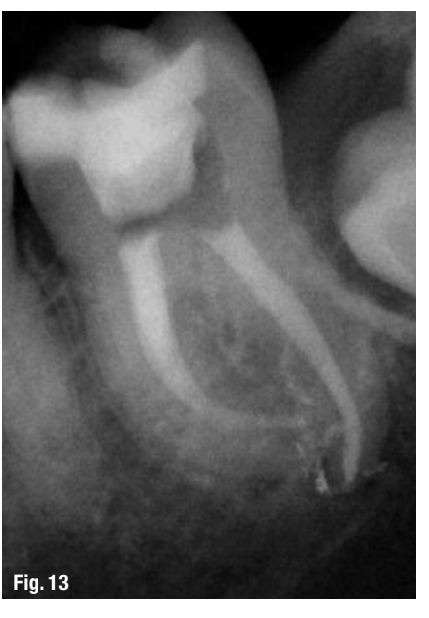
While this protocol is recommended for all endodontic treatments (Figs. 10–13), it is most valuable in the following clinical situations:
- infected cases with apical, lateral and/or furcal radiolucencies;
- retreatments with peri-apical periodontitis;
- acutely inflamed cases, especially those diagnosed with Cracked Tooth Syndrome;
- internal and external resorption;
- persistent infections not responding to conventional endodontic treatment; and
- unexplained, prolonged post-operative discomfort.
Summary
A root-canal cleaning, shaping and disinfection protocol has been described that maximises the removal of tissue, debris, smear layer and bacteria from root-canal systems. Utilising a combination of working width management with LightSpeed LSX instruments, high-volume apical negative pressure irrigation and evacuation with the EndoVac system and intra-canal laser therapy with radial-firing tips using the WaterlaseMD laser, the ability to eliminate bacteria from infected root-canal systems may soon be within our grasp.
References:
- Siqueira JF, Rocas IN: Clinical Implications and Microbiology of Bacterial Persistence after Treatment Procedures. J Endod. 2008;34(11):1291–1301.
- Gordon W, Atabakhsh VA, Meza F, Doms A, Nissan R, Rizoiu I, Stevens R: The antimicrobial efficacy of the erbium, chromium: yttrium-scandium-gallium-garnet laser with radial emitting tips on root canal dentin walls infected with Enterococcus faecalis. JADA. 2007;138(7):992–1002.
- Kouchi Y, Ninomiya J, Yasuda H, Fukui K, Moriyama T, Okamoto H: Location of streptococcus mutans in the dentinal tubules of open infected root canals. J Dent Res. 1980;59:2038–46.
- Haapasalo M, Orstavik D: In vitro infection and disinfection of dentinal tubules. J Dent Res. 1987;66(8):1375–9.
- Berutti E, Marini R,Angeretti A: Penetration ability of different irrigants into dentinal tubules. J Endod. 1997;23:725–7.
- Zou L, Shen Y, Li W, Haapasalo M: Penetration of Sodium Hypochlorite into Dentin. J Endod. 2010;36(5):793–6.
- Schoop U, Barylyak A, Goharkhay K, Beer F, Wernisch J, Georgopoulos A, Sperr W, Moritz A: The impact of a nerbium, chromium: yttrium-scandium-gallium-garnet laser with radial-firing tips on endodontic treatment. Lasers Med Sci. 2007;24(1):59–65.
- Allen F: In vivo study of apical cleaning. General Dentistry. 2007;449–56.
- Kerekes K, Tronstad L: Morphometric observations on root canals of human molars. J Endod. 1977;3:114–8.
- Wu MK, R’oris A, Barkis D, Wesselink P: Prevalence and extent of long oval canals in the apical third.OOO.2000;89(6):739–43.
- Chow TW: Mechanical effectiveness of root canal irrigation. JEndod. 1983;9(11):475–8.
- Tay FR, Gu L, Schoeffel GL, Wimmer C, Susin L, Zhang K, Arun SN, Kim J, Looney JW, Pashley DJ: Effect of Vapor Lock on Root Canal Debridement by using a Side-vented Needle for Positive-pressure Irrigant Delivery. J Endod. 2010;36(4):745–50.
- Schoeffel J: The EndoVac Method of Endodontic Irrigation, Part 2–Efficacy. Dentistry Today. 2008;27(1).
- Nielsen BA, Baumgartner JC:Comparison of the EndoVac System to Needle Irrigation of Root Canals. J Endod. 2007;33(5):611–5.
- Prashanth VS: Evaluation of New System for Root Canal Irrigation to Conventional: An ExVivo Study. Discus Dental, Culver City, CA: The EndoFiles Newsletter. 2008.
- Sung E, Rankin DD, Rizoiu I, Chueh P: Biolase Technology. un- published study. 2008.
- Peters O, Barbakow F: Effects of Irrigation on Debris and Smear Layer on Canal Walls:A Scanning Electron Microscopic Study. J Endod. 2000;26(1):6–10.
